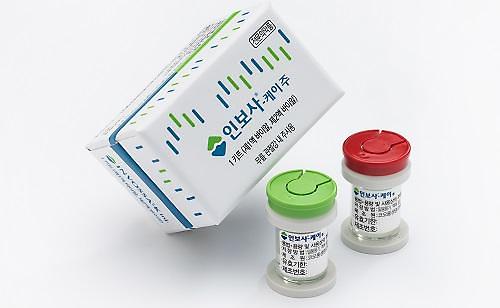
[Courtesy of Kolon Life Science]
SEOUL -- The U.S subsidiary of Kolon Life Science, a biosimilar arm of South Korea's Kolon Group, suspended patient recruitment for clinical trials to hold negotiations with the U.S. Food and Drug Administration after Invossa, the world's first cell-mediated gene therapy for degenerative arthritis, exposed a research mistake.
Kolon TissueGene said in a regulatory filing on Monday that it has started consultations with the American agency about the ingredients of Invossa. The company had been allowed to conduct phase 3 clinical trials involving patients diagnosed with knee osteoarthritis at 60 American hospitals.
Sales and distribution of Invossa were suspended in South Korea Sunday after Kolon TissueGene found during U.S. clinical trials that a key ingredient was different from that described in the data submitted at the time of permission in South Korea. The South's Ministry of Food and Drug Safety said it would conduct an inspection, although there was no concern about safety right now.
Kolon Life apologized for having used a wrong name, saying that there has been no change in the constituents of Invossa, which used the combination of human chondrocytes and transfected cells with TGF-β1 genes.
"The name of the cell has changed, not the constituents of Invossa," Kolon Life managing director Yoo Soo-hyun told reporters, adding Invossa has no safety and efficacy problems. He said confusion was caused by a different level of technology in analysis.
When analyzed for transfected cell characteristics in 2004, it was identified as chondrocytes because of the manifestation of cartilage cells, but the latest STR genetic test showed it was confirmed to be named differently. Invossa has been administered to 3,548 people for 11 years, but the company claimed to have discovered no serious side effects.
Kolon Life has described Invossa as a game changer in the global degenerative joint disease market. The company claimed a single injection would grant more than two years of pain-free movement in the knee without the immediate need for surgery.
Shares of Kolon TissueGene and Kolon Life tumbled 29.9 percent Monday, reflecting investors concerns. KB Securities analyst Lee Tae-yong said Invossa's commercialization could be delayed because Kolon TissueGene should change the main ingredients listed in its application. "It will take a considerable amount of time to resume clinical trials."