[Courtesy of EDGC]
SEOUL -- A South Korea-based healthcare company engaged in genetic analysis has introduced a liquid biopsy brand using techniques that can be used for the early detection of cancer and help doctors pick the right target drugs.
A liquid biopsy is the sampling and analysis technique for non-solid biological tissue such as blood. The technique is mainly used as a diagnostic and monitoring tool for diseases such as cancer. Because the liquid biopsy is almost non-invasive, it can be carried out more frequently than a biopsy that analyzes tissue samples removed from a tumor or a target body area.
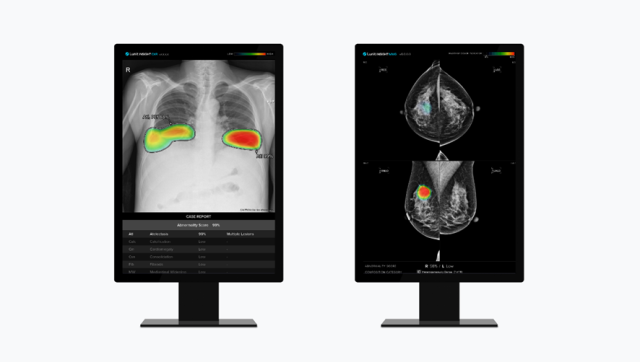
Eone Diagnomics Genome Center (EDGC) said in a statement that the company has launched "ONCOCATCH," a liquid biopsy brand. Doctors can use the brand to analyze patients' blood samples to detect cell-free DNA (cfDNA) and circulating tumor DNA (ctDNA) which are indicators used for the early detection of cancer.
The brand has four pipelines -- "ONCOCATCH-S," "ONCOCATCH-E," "ONCOCATCH-CDx," and "ONCOCATCH-M". ONCOCATCH-S is optimized for the early detection of lung cancer while artificial intelligence-based ONCOCATCH-E has the capability to diagnose the 10-most frequent cancers including colorectal cancer, lung cancer, breast cancer, and stomach cancer in early stages.
The ONCOCATCH-E diagnosis program can also pinpoint the location of cancer using AI-based algorithms. EDGC said that its ONCOCATCH-E is one of the top liquid biopsy brands in Asia.
The ONCOCATCH-CDx brand helps doctors pick the right treatment for patients and ONCOCATCH-M is a premium brand used for monitoring the reoccurrence and metastasis of cancer. The monitoring brand of ONCOCATCH can also analyze the efficacy of cancer treatments.
"OCOCATCH is capable of finding out differences in individuals' DNA and analyze a variety of information for the early detection of cancer without tissue samples," EDGC CEO Lee Min-seob was quoted as saying.
In March 2021, the Ministry of Food and Drug Safety certified that EDGC's ONCOCATCH brand complies with good manufacturing practices (GMPs) of in-vitro diagnostic devices. When EDGC receives state approval for commercialization, ONCOCATCH will be operated as a health check service program in the second half of 2022.