[Courtesy of ETRI]
Polymerase chain reaction (PCR) is a test method that is used to make millions of copies of a specific DNA sample to allow researchers to amplify a very small sample of DNA. Amplified DNA is large enough to be studied in detail. Quantitative PCR (qPCR), also known as real-time PCR, allows the quantification and detection of a specific DNA sequence in real-time. qPCR requires at least 20 microliters of swabbed specimens to get an accurate test result. If the amount of the right sample is too small, tests are likely to come up with "false negative" results because the amplified DNA will not meet the minimum requirements. qPCR is considered an analog method.
Digital PCR (dPCR) is a more accurate version of the PCR method. dPCR uses ultra-tiny droplets of two nanoliters that contain the sample DNA. A component called a fluorescence droplet reader is used to shoot laser beams into nano-sized liquid ball samples for analysis. dPCR can come up with accurate test results even when the amount of the target sample is very small. However, dPCR test equipment is more expensive and it costs more to operate than qPCR equipment.
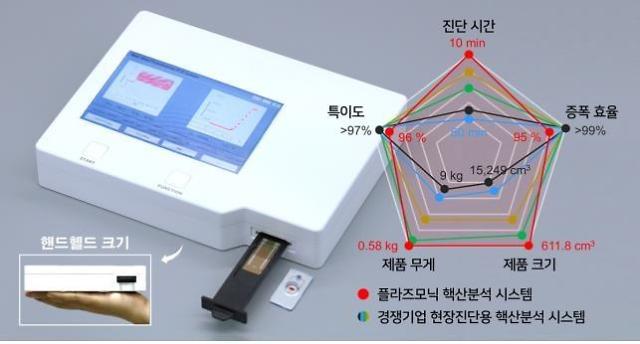
The Electronics and Telecommunications Research Institute (ETRI) said that its research team succeeded in the localization of a fluorescence droplet reader module that uses optical fibers with improved signal quality. Researchers said the newly-developed module is about half the price of conventional optical components used in dPCR equipment.
"Because the localized component will lower the manufacturing costs of dPCR test equipment, the distribution will be accelerated so that small-sized labs can use the new gear without much stress," ETRI researcher Kim Jin-tae told Aju Business Daily on March 3. He also said that the development of the integrated module will eventually lead to the miniaturization of the equipment. Current PCR test machines are very bulky. Test samples are collected from local screen centers and health clinics and carried to PCR labs for testing.
During the last week of February, South Korea saw a daily average of more than 170,000 people infected with COVID-19. About 98.8 percent of the detected virus was analyzed as the omicron variant, which is transmissible as highly contagious diseases such as measles. The explosive increase in the number of the infected has led researchers and health officials to seek more accurate test methods. Currently, the government prompts only those who received positive results from quick antigen tests to receive PCR tests to ease the workload of test labs.
However, the Korean Society for Laboratory Medicine had warned that the current COVID-19 test procedures that rely heavily on quick antigen test kits may bring a grievous effect because the kits have an accuracy of about 50 percent when the specimen is collected by a professional medical lab specialist.
If commercialized, the module developed by ETRI could provide means of highly accurate and affordable PCR tests to precisely track the spread of the infectious virus. The component is a chipset module integrated with a droplet generator, temperature control device, and a fluorescence reader. Because the module houses multiple devices, the production cost and time will be reduced.
[This article was contributed by Aju Business Daily intern reporter Lee So-lee]